Research
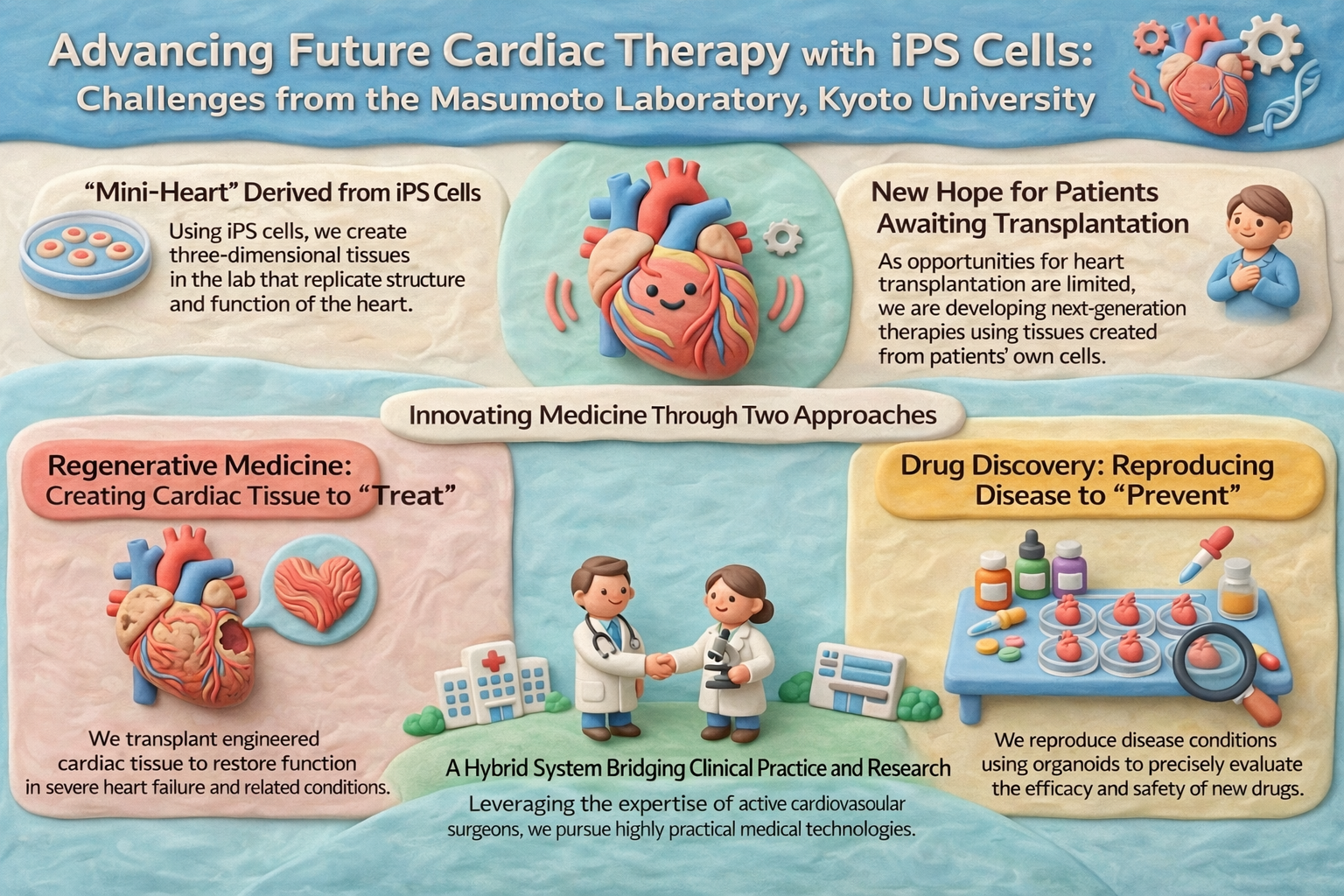
We aim to realize next-generation cardiovascular medicine based on human iPS cells and three-dimensional tissue engineering, with core strengths in regenerative medicine, disease modeling and drug discovery, and organoid maturation.
Three Core Research Areas
Building on human iPS cell and cardiac organoid technologies, we advance regenerative medicine, drug discovery, and organoid maturation in an integrated manner.
Regenerative Medicine
We develop transplantable cardiac tissues and vascularized constructs to establish new therapies for severe cardiovascular diseases.
Disease Modeling & Drug Discovery
Using disease models and heart-on-a-chip platforms, we investigate pathophysiology and evaluate drug efficacy and safety.
Organoid Maturation
We enhance physiological function through physical training approaches such as electrical and mechanical stimulation.
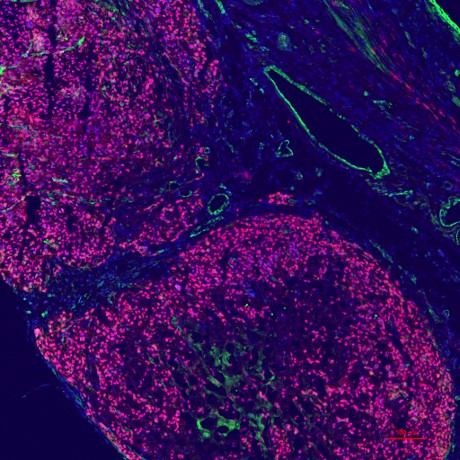
1. Regenerative Medicine
Our laboratory conducts a wide range of studies in regenerative medicine, with a particular focus on developing treatments for cardiac disease using iPS cells. We use multiple cardiovascular cell types derived from iPS cells, including cardiomyocytes, to explore new therapeutic strategies for myocardial infarction, heart failure, and related conditions.
In particular, we actively pursue organoid-based research grounded in tissue engineering approaches using iPS cells. By constructing organoids that recapitulate cardiac structure and function, we aim to elucidate mechanisms of cardiac regeneration and advance toward “true” regenerative medicine that restores myocardial function through tissue regeneration.
We also investigate the application of biomaterials to enhance therapeutic efficacy. These efforts are expected to support future clinical applications in cardiac regenerative medicine.
References
2. Disease Modeling and Drug Discovery
We use iPS cells and organoid technologies to recreate a variety of disease models and explore new opportunities for drug discovery. In particular, we focus on cardiovascular disease models to better understand disease mechanisms and develop novel therapeutic strategies.
We are also advancing drug discovery research using Organ-on-a-Chip (OoC) technology. By constructing heart-on-a-chip systems that mimic cardiac and vascular structures, we analyze intercellular interactions and drug responses under disease conditions.
This technology is also expected to serve as an alternative to animal experimentation and to facilitate the development of more human-relevant therapeutic approaches. Our work in disease modeling and drug discovery aims to deepen mechanistic understanding and enable innovative treatments through both regenerative medicine and pharmacological research.
References

3. Organoid Maturation
We aim to promote the maturation of iPS cell-derived organoids so that they more closely approximate physiological tissue states. Organoids are three-dimensional cellular assemblies that self-organize to mimic the structure of a specific organ, but insufficient maturation remains a major challenge for both regenerative medicine and disease modeling.
To address this, we apply “physical training” approaches, including mechanical and electrical stimulation. In cardiac organoids, mechanical loading that promotes contractile activity is used to enhance the maturation of cardiomyocyte function. These stimuli strengthen contractility and electrophysiological activity, enabling the generation of organoids with more realistic cardiac properties.
We also investigate the molecular mechanisms that govern organoid maturation and explore ways to regulate these processes. For example, we study activation of specific transcription factors and intercellular signaling pathways that promote maturation. Improving cell–cell interactions within organoids is another important strategy for enhancing maturity.
To evaluate mature organoids, we measure contractile force, electrophysiological activity, and metabolic function. Based on these assessments, we seek to identify the most effective stimuli and culture conditions that maximize physiological performance.
Our work on organoid maturation is expected to make major contributions to improving the precision of regenerative therapies and the reproducibility of disease models, ultimately opening the way toward more practical clinical applications.